GenoType MTBDRplus 2.0 Rifampicin + Isoniazid Resistance PCR CE/IVD
Catalog Number: HL-30412A, Size: 12 tests
Catalog Number: HL-30496A, Size: 96 tests
Molecular genetic assay for identification of resistance to rifampicin and/or isoniazid of Mycobacterium tuberculosis complex
Flyer, Questions? Contact us!
X
You are enquiring about GenoType MTBDRplus 2.0 Rifampicin + Isoniazid Resistance PCR CE/IVD
The emergence and spread of multidrug-resistant tuberculosis (MDR-TB) and extensively drug-resistant tuberculosis (XDR-TB) are a major medical and public problem threatening the global health.
MDR-TB is caused by mycobacteria which are at least resistant to the two most powerful first-line anti-TB drugs rifampicin and isoniazid. Conventional methods for mycobacteria culture and drug susceptibility testing are slow and elaborate, requiring sequential procedures for the diagnosis. During this time patients may be treated inappropriately, drug resistant strains may continue to spread, and amplification of resistance may occur. Therefore rapid diagnosis and identification of MDR-TB strains are prerequisites for the worldwide fight against TB.
The GenoType MTBDRplus enables a rapid result from pulmonary patient specimen and from culture material. Also for diagnosing patients after treatment failure and relapse, with unknown anamnesis and originating from high prevalence areas of MDR-TB as well as for diagnosing patients in high prevalence TB countries and high burden MDR-TB regions the use of GenoType MTBDRplus is reasonable. Finally the test can also be applied for screening purposes to develop country-specific TB action plans.
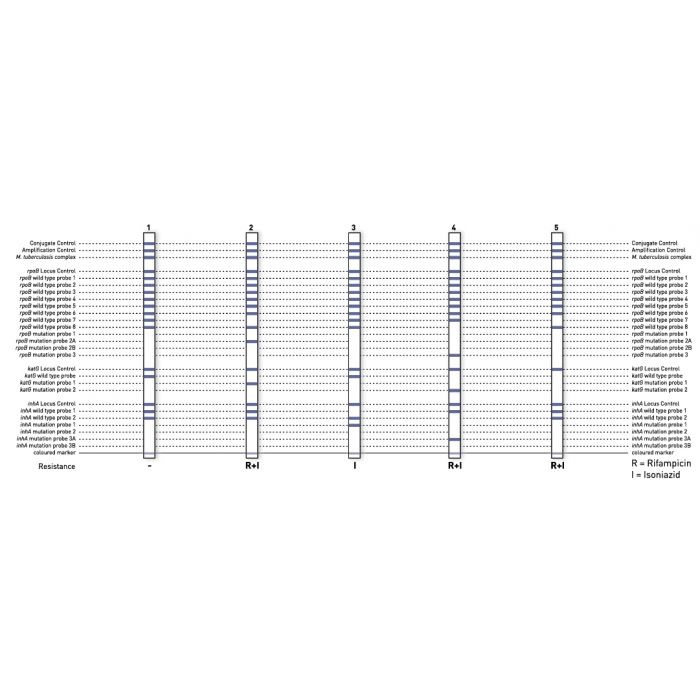
The identification of rifampicin resistance is enabled by the detection of the most significant mutations of the rpoB gene (coding for the β-subunit of the RNA polymerase). For testing the high level isoniazid resistance, the katG gene (coding for the catalase peroxidase) is examined and for testing the low level isoniazid resistance, the promoter region of the inhA gene (coding for the NADH enoyl ACP reductase) is analyzed.
Your benefits with GenoType MTBDRplus
- Can be performed from pulmonary patient specimen and from culture material.
- Results are obtained in 5hrs compared to 1 to 2 months with conventional methods.
- Allows early, appropriate treatment, which reduces transmission and spread of MDR-TB.
Molecular genetic assay for identification of resistance to rifampicin and/or isoniazid of Mycobacterium tuberculosis complex
Starting material:
Pulmonary specimens and/or liquid/solid culture samples
DNA Isolation:
GXT DNA/RNA Extraction (with GenoXtract®) or manually with GenoLyse®.
Flyer
Mycobacteria
Early Diagnosis
Facts About Resistance
Literature:
J Clin Microbiol. 2012 Apr;50(4):1264-9. doi: 10.1128/JCM.05903-11. Epub 2012 Feb 1.
First evaluation of an improved assay for molecular genetic detection of tuberculosis as well as rifampin and isoniazid resistances.
Crudu V, Stratan E, Romancenco E, Allerheiligen V, Hillemann A, Moraru N.
Microbiology & Morphology Laboratory Phthisiopneumology Institute, Chisinau, Moldova. valeriu.crudu@gmail.com
Abstract
The commercially available line probe assay MTBDRplus 2.0 (Hain Lifescience, Nehren, Germany) was evaluated for its ability to detect Mycobacterium tuberculosis complex (MTBC) and mutations conferring resistance to rifampin (RMP) and isoniazid (INH) directly in smear-negative and smear-positive pulmonary clinical specimens under routine laboratory conditions. A total of 348 samples originating from Moldova, a high-incidence country for tuberculosis (TB), were investigated. Two hundred fifty-seven (73.9%) were smear negative, 12 samples were excluded, and 81 (23.3%) were smear positive. Two DNA extraction methods were applied. Compared to culture and clinical data as the reference standard (adapted from Vadwai V et al., J. Clin. Microbiol. 49:2540-2545, 2011), overall sensitivity and specificity were 87.6 and 99.2%, respectively. One hundred four of the 257 smear-negative samples turned out to be culture positive, and 20 were MTBC culture negative but were positive based on clinical symptoms. The combined sensitivity and specificity in the subgroup of smear-negative samples were calculated to be 79.8 and 99.2%, respectively. MTBDRplus 2.0 detected RMP and INH resistance with sensitivity and specificity of 94.3 and 96.0%, respectively. In conclusion, the MTBDRplus 2.0 assay is a rapid and highly sensitive test for the detection of M. tuberculosis strains from smear-positive and -negative clinical specimens and provides additional information on RMP and INH resistance status, which can easily be included in routine laboratory work flow.
J Clin Microbiol. 2012 Nov;50(11):3712-6. doi: 10.1128/JCM.01958-12. Epub 2012 Sep 12.
The diagnostic performance of the GenoType MTBDRplus version 2 line probe assay is equivalent to that of the Xpert MTB/RIF assay.
Barnard M, Gey van Pittius NC, van Helden PD, Bosman M, Coetzee G, Warren RM.
Source
National Health Laboratory Service, Tuberculosis Reference Laboratory, Green Point, Cape Town, South Africa.
Abstract
Molecular diagnostics for Mycobacterium tuberculosis have recently been endorsed by the World Health Organization. The Xpert MTB/RIF assay was endorsed for use on patient material, regardless of smear gradation, while the GenoType MTBDRplus (version 1) has been limited for use on smear-positive patient material. In this study, we evaluated the diagnostic performance of the Xpert MTB/RIF and GenoType MTBDRplus (version 2) assays on smear-positive and smear-negative patient specimens submitted to a high-throughput diagnostic laboratory. A total of 282 consecutive specimens were subjected to the two new molecular assays, and their performance characteristics were assessed relative to the routine diagnostic standard. Both assays showed similar diagnostic performance characteristics. The sensitivities of the GenoType MTBDRplus (v2.0) and Xpert MTB/RIF assays for the detection of culture-positive M. tuberculosis were 73.1% and 71.2%, respectively, while the specificities of both assays were 100%. Both assays were able to diagnose the presence of M. tuberculosis in 57 to 58% of smear-negative cases, suggesting that the performance characteristics were dependent on bacillary load. The detection of M. tuberculosis in culture-negative specimens confirmed that molecular assays should not be used for treatment monitoring. The sensitivity and specificity for rifampin resistance detection were 100% in both assays; however, the GenoType MTBDRplus (v2.0) assay provided additional information on isoniazid susceptibility. The GenoType MTBDRplus (v2.0) assay will complement the Xpert MTB/RIF screening assay by validating rifampin susceptibility and providing information on isoniazid susceptibility. In addition, the GenoType MTBDRplus (v2.0) assay will provide pharmacogenetic information that may be critical in guiding appropriate treatment.
MDR-TB is caused by mycobacteria which are at least resistant to the two most powerful first-line anti-TB drugs rifampicin and isoniazid. Conventional methods for mycobacteria culture and drug susceptibility testing are slow and elaborate, requiring sequential procedures for the diagnosis. During this time patients may be treated inappropriately, drug resistant strains may continue to spread, and amplification of resistance may occur. Therefore rapid diagnosis and identification of MDR-TB strains are prerequisites for the worldwide fight against TB.
The GenoType MTBDRplus enables a rapid result from pulmonary patient specimen and from culture material. Also for diagnosing patients after treatment failure and relapse, with unknown anamnesis and originating from high prevalence areas of MDR-TB as well as for diagnosing patients in high prevalence TB countries and high burden MDR-TB regions the use of GenoType MTBDRplus is reasonable. Finally the test can also be applied for screening purposes to develop country-specific TB action plans.
The identification of rifampicin resistance is enabled by the detection of the most significant mutations of the rpoB gene (coding for the β-subunit of the RNA polymerase). For testing the high level isoniazid resistance, the katG gene (coding for the catalase peroxidase) is examined and for testing the low level isoniazid resistance, the promoter region of the inhA gene (coding for the NADH enoyl ACP reductase) is analyzed.
Your benefits with GenoType MTBDRplus
- Can be performed from pulmonary patient specimen and from culture material.
- Results are obtained in 5hrs compared to 1 to 2 months with conventional methods.
- Allows early, appropriate treatment, which reduces transmission and spread of MDR-TB.
Molecular genetic assay for identification of resistance to rifampicin and/or isoniazid of Mycobacterium tuberculosis complex
Starting material:
Pulmonary specimens and/or liquid/solid culture samples
DNA Isolation:
GXT DNA/RNA Extraction (with GenoXtract®) or manually with GenoLyse®.
Flyer
Mycobacteria
Early Diagnosis
Facts About Resistance
Literature:
J Clin Microbiol. 2012 Apr;50(4):1264-9. doi: 10.1128/JCM.05903-11. Epub 2012 Feb 1.
First evaluation of an improved assay for molecular genetic detection of tuberculosis as well as rifampin and isoniazid resistances.
Crudu V, Stratan E, Romancenco E, Allerheiligen V, Hillemann A, Moraru N.
Microbiology & Morphology Laboratory Phthisiopneumology Institute, Chisinau, Moldova. valeriu.crudu@gmail.com
Abstract
The commercially available line probe assay MTBDRplus 2.0 (Hain Lifescience, Nehren, Germany) was evaluated for its ability to detect Mycobacterium tuberculosis complex (MTBC) and mutations conferring resistance to rifampin (RMP) and isoniazid (INH) directly in smear-negative and smear-positive pulmonary clinical specimens under routine laboratory conditions. A total of 348 samples originating from Moldova, a high-incidence country for tuberculosis (TB), were investigated. Two hundred fifty-seven (73.9%) were smear negative, 12 samples were excluded, and 81 (23.3%) were smear positive. Two DNA extraction methods were applied. Compared to culture and clinical data as the reference standard (adapted from Vadwai V et al., J. Clin. Microbiol. 49:2540-2545, 2011), overall sensitivity and specificity were 87.6 and 99.2%, respectively. One hundred four of the 257 smear-negative samples turned out to be culture positive, and 20 were MTBC culture negative but were positive based on clinical symptoms. The combined sensitivity and specificity in the subgroup of smear-negative samples were calculated to be 79.8 and 99.2%, respectively. MTBDRplus 2.0 detected RMP and INH resistance with sensitivity and specificity of 94.3 and 96.0%, respectively. In conclusion, the MTBDRplus 2.0 assay is a rapid and highly sensitive test for the detection of M. tuberculosis strains from smear-positive and -negative clinical specimens and provides additional information on RMP and INH resistance status, which can easily be included in routine laboratory work flow.
J Clin Microbiol. 2012 Nov;50(11):3712-6. doi: 10.1128/JCM.01958-12. Epub 2012 Sep 12.
The diagnostic performance of the GenoType MTBDRplus version 2 line probe assay is equivalent to that of the Xpert MTB/RIF assay.
Barnard M, Gey van Pittius NC, van Helden PD, Bosman M, Coetzee G, Warren RM.
Source
National Health Laboratory Service, Tuberculosis Reference Laboratory, Green Point, Cape Town, South Africa.
Abstract
Molecular diagnostics for Mycobacterium tuberculosis have recently been endorsed by the World Health Organization. The Xpert MTB/RIF assay was endorsed for use on patient material, regardless of smear gradation, while the GenoType MTBDRplus (version 1) has been limited for use on smear-positive patient material. In this study, we evaluated the diagnostic performance of the Xpert MTB/RIF and GenoType MTBDRplus (version 2) assays on smear-positive and smear-negative patient specimens submitted to a high-throughput diagnostic laboratory. A total of 282 consecutive specimens were subjected to the two new molecular assays, and their performance characteristics were assessed relative to the routine diagnostic standard. Both assays showed similar diagnostic performance characteristics. The sensitivities of the GenoType MTBDRplus (v2.0) and Xpert MTB/RIF assays for the detection of culture-positive M. tuberculosis were 73.1% and 71.2%, respectively, while the specificities of both assays were 100%. Both assays were able to diagnose the presence of M. tuberculosis in 57 to 58% of smear-negative cases, suggesting that the performance characteristics were dependent on bacillary load. The detection of M. tuberculosis in culture-negative specimens confirmed that molecular assays should not be used for treatment monitoring. The sensitivity and specificity for rifampin resistance detection were 100% in both assays; however, the GenoType MTBDRplus (v2.0) assay provided additional information on isoniazid susceptibility. The GenoType MTBDRplus (v2.0) assay will complement the Xpert MTB/RIF screening assay by validating rifampin susceptibility and providing information on isoniazid susceptibility. In addition, the GenoType MTBDRplus (v2.0) assay will provide pharmacogenetic information that may be critical in guiding appropriate treatment.
| Is Featured? | No |
|---|
Write Your Own Review