Gram-negative Nosocomial Agents I (Enterobacter spp., Acinetobacter baumannii and E. coli) lyophilised RTU qPCR CE/IVD
SKU: VIT-7041073
SKU: VIT-7042073 (high profile strip)
Size: 4x8-well strip
Real-time PCR kit for the qualitative detection and identification of Enterobacter spp., Acinetobacter baumannii and/or Escherichia coli species in clinical samples.
Datasheet
Questions? Contact us!
SKU: VIT-7042073 (high profile strip)
Size: 4x8-well strip
Real-time PCR kit for the qualitative detection and identification of Enterobacter spp., Acinetobacter baumannii and/or Escherichia coli species in clinical samples.
Datasheet
Questions? Contact us!
X
You are enquiring about Gram-negative Nosocomial Agents I (Enterobacter spp., Acinetobacter baumannii and E. coli) lyophilised RTU qPCR CE/IVD
Intended Use:
Vitassay qPCR Gram-negative Nosocomial Agents I allows the qualitative detection and differentiation of DNA from Enterobacter spp., Acinetobacter baumannii and Escherichia coli by Real-Time PCR in blood culture and swab samples from patients with suspect bacterial infection and/or multi-resistant infection or colonization and swabs specimens for epidemiological control by their healthcare professional. This product is intended to aid in the Enterobacter spp., Acinetobacter baumannii and Escherichia coli infections diagnosis, alongside the patient's clinical data, epidemiological risk factors, and other laboratory tests outcomes.
The kits include a positive and a negative control.
Identification of pathogen DNA or RNA occurs by the use of target specific primers ans fluorecent labeled hydrolysis probes which hybridize conserved regions in the genomes of the different pathogens.
Kit Content:
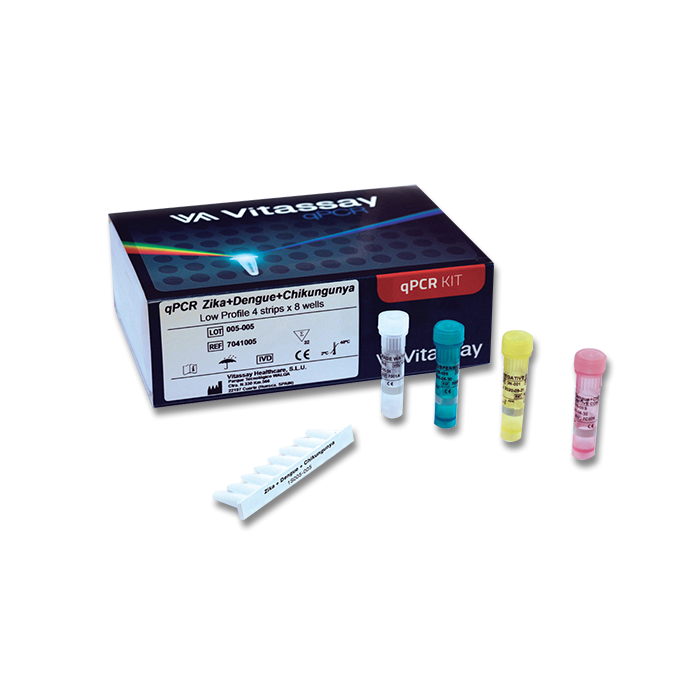
- Gram-negative I strips low/high profile : 4 x 8-well strip
- Gram-negative I Positive Control: 1 vials
- PCR grade water (white vial): 1 vial
- Resuspension Buffer (green vial): 1 vial x1,8 mL
- Negative control (yellow): 1 vial x 1 mL
- Optical caps: 4x8 cap strip
Kit Format:
qualitative
Vitassay qPCR Gram-negative Nosocomial Agents I allows the qualitative detection and differentiation of DNA from Enterobacter spp., Acinetobacter baumannii and Escherichia coli by Real-Time PCR in blood culture and swab samples from patients with suspect bacterial infection and/or multi-resistant infection or colonization and swabs specimens for epidemiological control by their healthcare professional. This product is intended to aid in the Enterobacter spp., Acinetobacter baumannii and Escherichia coli infections diagnosis, alongside the patient's clinical data, epidemiological risk factors, and other laboratory tests outcomes.
The kits include a positive and a negative control.
Identification of pathogen DNA or RNA occurs by the use of target specific primers ans fluorecent labeled hydrolysis probes which hybridize conserved regions in the genomes of the different pathogens.
Kit Content:
- Gram-negative I strips low/high profile : 4 x 8-well strip
- Gram-negative I Positive Control: 1 vials
- PCR grade water (white vial): 1 vial
- Resuspension Buffer (green vial): 1 vial x1,8 mL
- Negative control (yellow): 1 vial x 1 mL
- Optical caps: 4x8 cap strip
Kit Format:
qualitative
| Is Featured? | No |
|---|
Write Your Own Review